Welcome to our comprehensive infographics database, a curated collection of evidence-based visual guides designed to support best practices in laboratory safety, biosecurity, and public health protocols. Developed in collaboration with leading international health organizations including the CDC, WHO, NIOSH, and IATA, these infographics serve as essential reference materials for researchers, healthcare professionals, laboratory personnel, and public health practitioners.
Our database addresses critical areas of laboratory operations and safety compliance, from personal protective equipment standards and specimen collection protocols to advanced biosafety containment systems and emergency response procedures. Each infographic is meticulously crafted to translate complex regulatory guidelines and technical protocols into clear, actionable visual formats that enhance understanding and promote adherence to international safety standards.
The collection encompasses fundamental safety protocols essential for modern laboratory operations, including respiratory protection guidelines that ensure proper fit-testing compliance, specimen handling procedures that maintain sample integrity while protecting personnel, and biosafety containment verification systems critical for high-risk pathogen research. Additionally, our infographics cover infection control measures such as evidence-based hand hygiene protocols proven to reduce healthcare-associated infections by up to 50%, and chemical disinfection procedures that provide broad-spectrum antimicrobial efficacy against bacteria, viruses, fungi, and spores.
These visual resources are particularly valuable for training programs, laboratory certification processes, and regulatory compliance verification. They serve multiple audiences, from undergraduate students learning fundamental laboratory techniques to experienced researchers working with Risk Group 3 biological agents in BSL-3 facilities. The infographics are designed to be printed for quick reference in laboratory settings, integrated into training curricula, or used as standardized operating procedure supplements.
Our commitment to scientific accuracy ensures that each infographic undergoes rigorous review against current regulatory frameworks and incorporates the latest updates from authoritative sources. The visual design prioritizes clarity and accessibility while maintaining technical precision, making complex protocols easily understood and properly implemented across diverse laboratory environments.
Whether you are establishing new laboratory protocols, conducting safety training, preparing for regulatory inspections, or seeking quick reference materials for daily operations, this infographics database provides authoritative, up-to-date visual guidance that supports excellence in laboratory safety and public health practice. We continuously expand this collection to address emerging safety challenges and evolving regulatory requirements in the rapidly advancing fields of genomics, infectious disease research, and laboratory medicine.
Explore our collection to enhance your laboratory's safety culture, improve protocol compliance, and ensure that your team has access to the most current and authoritative guidance for safe, effective laboratory operations.
Essential information for laboratory safety and public health
| Brief Atlas: American Hantavirus Rodent Reservoirs
This comprehensive atlas provides essential identification and distribution information for rodent species that serve as natural reservoirs for hantaviruses across the Americas. Hantaviruses pose significant public health risks, causing hantavirus pulmonary syndrome (HPS) and hemorrhagic fever with renal syndrome (HFRS), with case fatality rates reaching up to 40%. The atlas features detailed species profiles, geographic distribution maps, and morphological characteristics of key reservoir hosts including deer mice (Peromyscus maniculatus), cotton rats (Sigmodon species), and rice rats (Oryzomys species). This resource is invaluable for public health professionals, field researchers, veterinarians, and laboratory personnel working in endemic areas, providing critical information for disease surveillance, risk assessment, and prevention strategies in both rural and periurban environments where human-rodent contact may occur.. |  |
| CDC and NIOSH approved facial hairstyles
This infographic, based on CDC and NIOSH guidelines, depicts facial hairstyles that are compatible with tight-fitting respirators. It identifies which beard and mustache styles are acceptable or prohibited based on their potential to interfere with the respirator seal. Personnel must ensure that facial hair does not compromise the respirator’s fit. Styles that cross or extend beneath the sealing surface are not allowed, while trimmed, non-interfering styles may be acceptable. Compliance with these grooming standards is required for all respirator users. |  |
| IATA Triple Packaging System
The IATA Triple Packaging System ensures the safe transport of infectious substances under IATA’s Dangerous Goods Regulations (DGR) and is widely adopted by the CDC for biosafety. Its objectives are to prevent leakage, protect personnel, and ensure compliance. The system includes a leak-proof primary container, a secondary protective layer, and a rigid outer container for impact resistance. This design enhances security by preventing exposure, reducing contamination risks, and preserving specimen integrity, making it a global standard in public health and laboratory transport. | 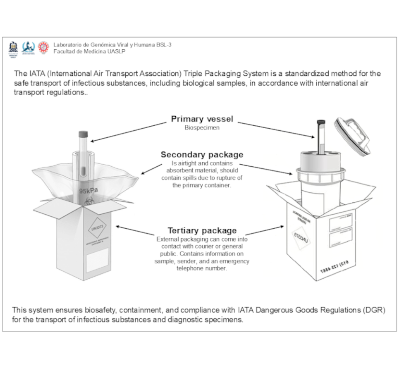 |
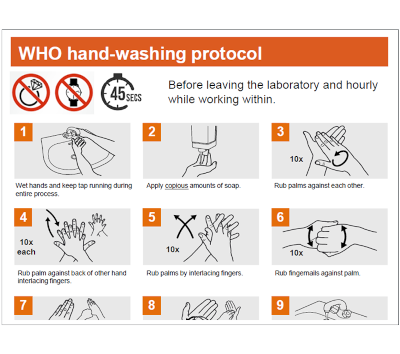
| World Health Organization (WHO) hand-washing protocol WHO hand hygiene protocol is a standardized five-moment approach for hand cleaning that prevents healthcare-associated infections and reduces pathogen transmission in clinical settings. The WHO strongly recommends this evidence-based protocol because proper hand hygiene reduces healthcare-associated infections by up to 50% when implemented correctly. The protocol involves alcohol-based hand rub or soap and water application at five critical moments: before patient contact, before aseptic procedures, after body fluid exposure risk, after patient contact, and after contact with patient surroundings. This systematic approach is essential for protecting patients, healthcare workers, and visitors from preventable infections in hospitals, clinics, and long-term care facilities worldwide. |
 |
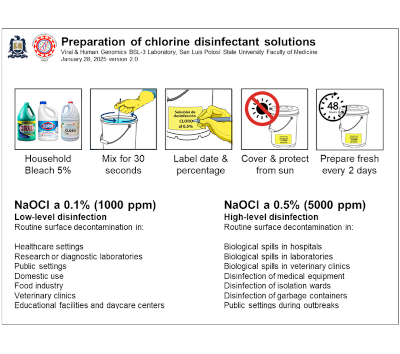
| Chlorine disinfectant solution preparation Chlorine disinfectant solution preparation is a standardized dilution process for creating effective antimicrobial solutions from concentrated sodium hypochlorite (bleach) for surface and equipment decontamination. The CDC and WHO recommend chlorine-based disinfectants because they provide broad-spectrum antimicrobial activity against bacteria, viruses, fungi, and spores, with working solutions ranging from 0.1% (1,000 ppm) for general surfaces to 0.5% (5,000 ppm) for blood spills. The preparation involves accurate dilution calculations and immediate use protocols since chlorine solutions lose potency over time when exposed to light, heat, and organic matter. This method is crucial for infection control, laboratory decontamination, and outbreak response in healthcare and research facilities.. |
 |
Laboratory procedures and operations
| Serum separation guide for clinical whole blood samples
Serum is the liquid component of blood obtained after clotting, commonly used for a wide range of diagnostic tests, including chemistry panels, immunoassays, and infectious disease serology. Proper serum separation is essential to ensure accurate and reliable laboratory results. After whole blood is collected in a clot-activator or whole blood VacutainerTM tube, the sample is allowed to clot and then centrifuged to isolate the clear, cell-free serum layer. This process removes cellular components that can interfere with analyte stability or skew test measurements. Consistent technique in clotting time, centrifugation conditions, and post-centrifugation handling helps maintain sample integrity, supporting high-quality diagnostics, timely clinical decisions, and effective patient care. | 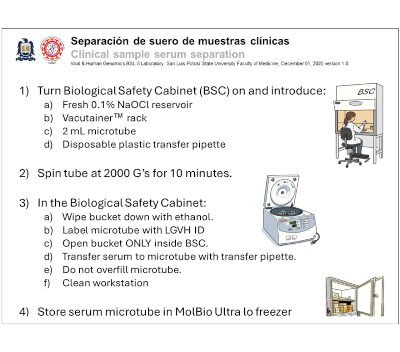 |
| Baseline serum processing
Baseline serum aliquots are a critical biosafety resource in BSL-2 and BSL-3 laboratories, providing a documented pre-exposure reference for laboratory personnel. In the event of a suspected laboratory-acquired infection, these aliquots enable retrospective serological comparison to confirm exposure, assess seroconversion, and support incident investigations. They also assist occupational health services in guiding timely medical evaluation, treatment, and long-term follow-up of affected staff. |  |
| Nasopharyngeal specimen collection guide
A nasopharyngeal biospecimen is a sample collected from the upper part of the throat behind the nose, widely used for detecting respiratory viruses like COVID-19 and influenza. The CDC recommends this biospecimen type because the nasopharynx harbors a high viral load, making it more sensitive and accurate than other sample types, such as oropharyngeal or anterior nasal swabs. The collection process involves inserting a swab deep into the nasal cavity to obtain mucus and cellular material for PCR or antigen-based testing. This method is crucial for early detection, outbreak control, and reducing transmission in both clinical and public health settings. |  |
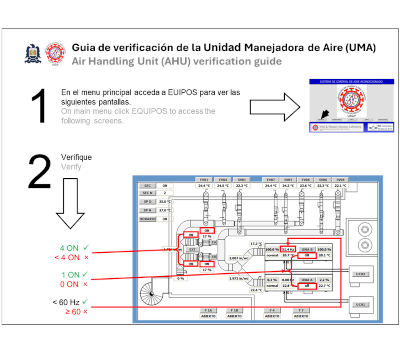
| Air Handling Unit (AHU) operational status verification A BSL-3 laboratory Air Handling Unit (AHU) operational status verification is a critical safety protocol that ensures proper containment of infectious agents through continuous monitoring of airflow parameters. The CDC and NIH guidelines mandate this verification because proper directional airflow and negative pressure differentials are essential for preventing the escape of potentially dangerous pathogens from the containment laboratory. The verification process involves real-time monitoring of air changes per hour (ACH), pressure differentials, HEPA filter integrity, and exhaust system performance to maintain the required inward airflow at all laboratory access points. This monitoring system is fundamental for personnel safety, environmental protection, and regulatory compliance in facilities handling Risk Group 3 biological agents. |
 |